Introducing 3:1 heat shrink tubing to have you labeling like a Pro
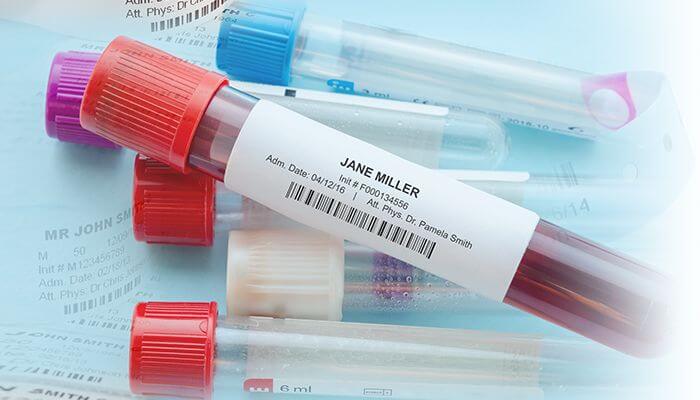
Slide Labeling Helps Reduce Misidentification Errors
According to the 2013 Deep Dive Analysis on Laboratory Testing study conducted by the ECRI Institute, specimen mislabeling is the leading cause of errors in the laboratory testing process. This includes a specimen with a label from a different patient, two contradictory labels, or a label that does not correspond with the order.
Of the nearly 2,500 patient safety events reviewed in the study, 31 percent were due to mislabeling. The study also states mislabeled specimens can lead to delays in the lab, such as the re-collection of samples, which can cause patient harm such as obtaining important diagnostic information for patient care.
To help prevent these types of errors, one of the Joint Commission’s 2014 National Patient Safety Goals for the Laboratory Accreditation Program is to “use at least two patient identifiers when providing laboratory services.”
Printing standardized bar code slide labels helps improve patient safety by providing positive patient identification. Handwritten or faulty labels that smear or fade can contribute to the misdiagnosis of patients and an increase in unnecessary costs linked to retesting. If a handwritten label is misread, patients can be put in danger and hospitals at risk for liability of misdiagnoses. Using bar coded labels eliminates handwritten inaccuracies to provide lab technicians with peace of mind that patient ID information is accurately printed in the lab.
Standardized bar code slide labels also comply with regulatory requirements, such as the Laboratory Accreditation Program from the Joint Commission and the AUTO12-A standard enacted by the Clinical and Laboratory Standards Institute (CLSI). Improving lab workflow efficiencies and performance are other benefits of bar coding, allowing technicians to spend more time on specimen testing to more quickly diagnose patients.
Current printer technology can allow lab technicians to “drop and print” a variety of label types without recalibrating in between. Using smart technology, these printers automatically sense the type and size of label media inserted, saving technicians’ time as there is no need for reformatting.
The media that comes with these printers should be resistant to oils, plasticizers, and water — all of which are common components found and used in a laboratory setting. Additionally, these labels should be made of durable materials able to withstand up to 200°F with adhesive strong enough that technicians do not have to worry about labels detaching from slides during testing and storage.






